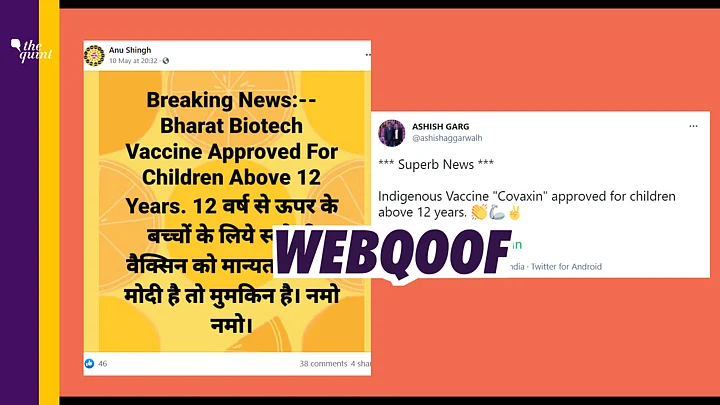
A claim going viral on the internet states that Bharat Biotech’s COVID-19 vaccine Covaxin has been approved for being used on children above the age of 12 years.
The claim comes as eminent scientists and doctors have warned of a third wave of coronavirus infections, that could possibly be worse for the unvaccinated population of those under the age of 18.
However, we found that Covaxin has not yet been approved for use on children above 12 years of age.
On 13 May, the Drugs Controller General of India (DCGI) accepted the recommendation of the Subject Expert Committee (SEC) and granted permission to conduct Phase II/III clinical trials of Bharat Biotech's COVID-19 vaccine, Covaxin, in the age group of 2-18 years.
WHAT WE FOUND
We looked up the website and social media handles of the Ministry of Health Affairs and Family Welfare (MoHFW), and didn't find the mention of any vaccine being approved for those under the age of 18 years.
India approved both Covaxin and Serum Institute of India-made Covishield vaccine on 3 January and phase one of inoculation drive began on 16 January for healthcare and front-line workers. The second and third phase of the vaccination drive included those above the age of 60, followed by all those above the age of 45 years.
Starting 1 May, everyone above the age of 18 have been allowed to take the vaccine. However, there was no mention of vaccinating anyone under 18.
The FAQ section on the health ministry's website also says, "The general practice is to first evaluate any new vaccine in older population and then age reduction is done to assess the safety and effectiveness in paediatric population. The currently available vaccines have not been evaluated in children so far. There are some clinical trials now underway to test the effectiveness and safety of the COVID-19 vaccines in children."
According to news reports, the Bharat Biotech’s vaccine got it approval to conduct trials on children above the age of 12 years on 4 January 2021.
A news report published on 13 May that said that the DCGI had granted permission to conduct Phase II/III clinical trials of Covaxin in the age group of 2-18 years. The trials will be conducted by Bharat Biotech in 525 healthy volunteers, the Centre said in a statement.
Evidently, Covaxin has not yet been approved for use on children above the age of 12 years. It is still in the clinical trial stage.
(Not convinced of a post or information you came across online and want it verified? Send us the details on WhatsApp at 9643651818, or e-mail it to us at webqoof@thequint.com and we'll fact-check it for you. You can also read all our fact-checked stories here.)